Technology
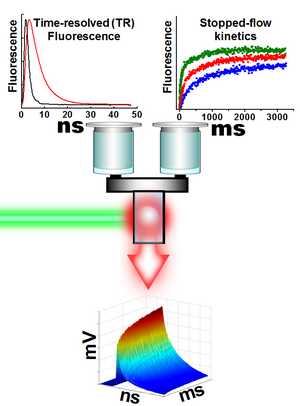
Time-resolved luminescence is an extremely popular technique in life science and biotechnology.
Using time-resolved resonance energy transfer in site-specific labeled protein samples scientists
can determine intra-molecular distances and their distributions. In this technique the decay of
luminescence is measured after a short excitation pulse. The Direct Waveform Recording is one of
the methods to measure time-resolved luminescence. In this method an electrical signal from an optical detector
is digitized by a fast analog-digital converter. This technique is often referred
to us as single pulse approach because
it lets one get information about whole decay using a single excitation shot. Because this process
is very fast and can be quickly repeated many times, it could be combined with one of the conventional kinetic
methods such as stopped-flow or flash-photolysis to measure the kinetics of biochemical reactions. The resulting method
is often called double kinetic because there are two kinetics that involved in it: kinetic of luminescence
decay and kinetic of biochemical reaction.
Double Kinetic Systems designs instruments for double kinetic experiments by merging Direct Waveform
Recording and stopped-flow methods in a single instrument. The biochemical reaction is initialized in
a stopped-flow apparatus by mixing two compounds. Start of data acquisition is synchronized with the end of
the stopped-flow syringe push. Luminescence is excited using high repetition rate microchip YAG laser.
Available wavelengths are 532 nm (2nd harmonic of YAG laser), 355 nm (3rd harmonic),
and 266 nm (4th harmonic).
After start of the data acquisition this instrument measures the entire luminescence decay for every excitation laser flash
so hundreds of decays per second are measured with sub-nanosecond time resolution in luminescence signal.
As a result, a sequence of time tagged waveforms is obtained. Delay between waveforms is calculated as the
difference between time tags. Waveforms in each sequence can be grouped and averaged in each group to reduce
time resolution in biochemical reaction time scale while increasing signal to noise ratio. Even higher signal
to noise ratio can be obtained by averaging data for several stopped-flow shots. As a result, one can get
not only distances between labels in studied protein by analyzing individual luminescence decays, but get
millisecond “motion picture” of its kinetic with possibility of detecting intermediate states.